Microbiology
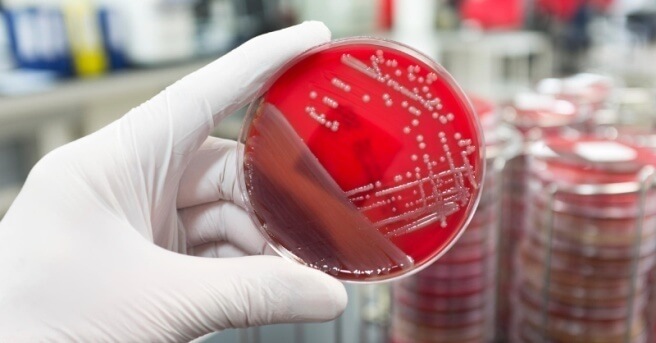
Microbiology testing services are a crucial requirement across many industries worldwide where products, processes and human health are at risk of being negatively affected by the presence and breeding of micro-organisms such as specific pathogens, bacteria, yeast and moulds.
Our laboratory offer microbiological testing across many industries including drugs and Pharmaceuticals Products, food, consumer products, cosmetics and beauty products, medical devices, drinking water etc.
From assessment of microbial contamination, quality control testing, shelf-life, our microbiology testing services can increase consumer confidence in your product, mitigate the negative effects of microbes in your production environment and safeguard your processes and personnel.
Microbial Limit Test:
The microbial limits test (MLT) are performed to determine whether a product complies with the specifications for microbial quality, and consists of two parts. The quantitative phase, Microbial Enumeration, determines the total number of aerobic organisms as well as a total yeast and mould count on a product. The qualitative phase of a microbial limits test is known as the Test for Specified Microorganisms. This test is designed to determine the presence or absence of specific objectionable organisms in a product.
Available Pharmaceutical Microbial Limits Testing Services
MLT Method Suitability Test
Microbial Enumeration (Total Aerobic Microbial Count and Total Combined Yeasts and Mold Count)
Screening Test for Specified Microorganisms:
P. aeruginosa
E. coli
S. aureus
Salmonella
Clostridium
Sterility Testing:
Sterility Testing is defined as a testing which confirms that products are free from the presence of viable microorganisms. Sterility testing is very important for medical devices, pharmaceuticals, preparations, tissue materials and other materials that claim to be sterile or free from viable microorganisms.
Using validated methods, products exposed to a sterilization process are tested to confirm the continued effectiveness of the selected method of sterilization. The product or representative product challenge devices (PCDs) are placed in a nutrient broth and placed into incubation at the appropriate temperature and time frame for the method selected.
Incubation parameters generally recommended:
Direct Transfer
· Biological indicators; 7 days at 32.5 ±2.5° C
· Product; 14 days at 22.5 ±2.5° C & 32.5 ±2.5° C
· Product (gamma verification); 14 days at 30 ±2° C
Membrane Filtration
· Standard method or closed system Millipore Steritest
· 14 days at 22.5 ±2.5° C & 32.5 ±2.5° C
Bacterial Endotoxin (BET) / LAL
This in vitro technique tests for the presence of bacterial endotoxin in or on medical products which will come in contact with the cardiovascular or lymphatic systems or cerebrospinal fluid. It is used to support a label claim of “non-pyrogenic” or “pyrogen free”. The Inhibition Enhancement test validates minimal product interference with the BET.
The In-Process LAL test is typically used to monitor raw material/sub-assemblies/process material (e.g. water) for endotoxin. A quantitative LAL test estimates the level of endotoxin in/on a product when found to be present in a gel clot limit test.
Microorganism Analysis
FDA and ISO guidance recommend characterization of the microorganisms recovered from a bioburden test or by environmental monitoring. The characterization allows for detection of change to the normal bio-load of a product, particularly if the bioburden data is used to establish a sterilization process.
SBTL offers the following to provide this information:
· Gram Stain
· Cell Morphology
· Colony Morphology
· Selective culturing for specific organisms
USP Antimicrobial Effectiveness Test
This test determines the effectiveness of an antimicrobial preservative over the life of a product. This effectiveness must be demonstrated for all injections packaged in multiple-dose containers and multiple-dose topical and oral dosage form.
USP Microbial Limits
When microbiological quality of a material is required in an individual monograph, this testing estimates quantitative levels of aerobic organisms to determine whether the material or formula meets the specifications. Five organisms are indicated for use in this testing: Staphylococcus aureus, Pseudomonas aeruginosa, Bacillus subtilis, Candida albicans, and Aspergillus niger.
Typically, these tests are performed by a membrane filtration method or the plate count method. A validation must be performed to assure the test method’s capability to detect microorganisms if present. Any change in a product’s formulation would require revalidation of the test method.
Bioburden
Definition: the population of viable microorganisms on or in a product and/or sterile barrier system.
An important part of the manufacturing process for medical devices is determining the level and nature of viable microorganisms that are generated as a result of the process. By developing and following a routine, documented sampling plan, you are assured that your process remains under control.
At SBTL, all bioburden testing is performed in a clean room, using a validated method most suited to your product following recovery methods outlined in ANSI/AAMI/ISO 11737-1. Various methods are considered; shaking, ultrasonication, stomaching, fluid pathway, membrane filtration, disintegration by blending, or swabbing are some of the methods that are reviewed to determine the optimal method for an individual product.
For medical devices, the most commonly requested information is:
· Aerobic Total Count
· Aerobic Total and Spore Count
· Mold and Fungi Total Count
· Anaerobic Total Count
· Anaerobic Total and Spore Count
Additionally, we service the personal care industry, performing bioburden estimation or screening for specific contaminants on products such as wipes, soaps, and lotions. To monitor process water and/or water systems, a Heterotrophic Plate Count and Total Coliform Count are suggested.
Microbiology

Microbiology testing services are a crucial requirement across many industries worldwide where products, processes and human health are at risk of being negatively affected by the presence and breeding of micro-organisms such as specific pathogens, bacteria, yeast and moulds.
Our laboratory offer microbiological testing across many industries including drugs and Pharmaceuticals Products, food, consumer products, cosmetics and beauty products, medical devices, drinking water etc.
From assessment of microbial contamination, quality control testing, shelf-life, our microbiology testing services can increase consumer confidence in your product, mitigate the negative effects of microbes in your production environment and safeguard your processes and personnel.
Microbial Limit Test:
The microbial limits test (MLT) are performed to determine whether a product complies with the specifications for microbial quality, and consists of two parts. The quantitative phase, Microbial Enumeration, determines the total number of aerobic organisms as well as a total yeast and mould count on a product. The qualitative phase of a microbial limits test is known as the Test for Specified Microorganisms. This test is designed to determine the presence or absence of specific objectionable organisms in a product.
Available Pharmaceutical Microbial Limits Testing Services
MLT Method Suitability Test
Microbial Enumeration (Total Aerobic Microbial Count and Total Combined Yeasts and Mold Count)
Screening Test for Specified Microorganisms:
P. aeruginosa
E. coli
S. aureus
Salmonella
Clostridium
Sterility Testing:
Sterility Testing is defined as a testing which confirms that products are free from the presence of viable microorganisms. Sterility testing is very important for medical devices, pharmaceuticals, preparations, tissue materials and other materials that claim to be sterile or free from viable microorganisms.
Using validated methods, products exposed to a sterilization process are tested to confirm the continued effectiveness of the selected method of sterilization. The product or representative product challenge devices (PCDs) are placed in a nutrient broth and placed into incubation at the appropriate temperature and time frame for the method selected.
Incubation parameters generally recommended:
Direct Transfer
· Biological indicators; 7 days at 32.5 ±2.5° C
· Product; 14 days at 22.5 ±2.5° C & 32.5 ±2.5° C
· Product (gamma verification); 14 days at 30 ±2° C
Membrane Filtration
· Standard method or closed system Millipore Steritest
· 14 days at 22.5 ±2.5° C & 32.5 ±2.5° C
Bacterial Endotoxin (BET) / LAL
This in vitro technique tests for the presence of bacterial endotoxin in or on medical products which will come in contact with the cardiovascular or lymphatic systems or cerebrospinal fluid. It is used to support a label claim of “non-pyrogenic” or “pyrogen free”. The Inhibition Enhancement test validates minimal product interference with the BET.
The In-Process LAL test is typically used to monitor raw material/sub-assemblies/process material (e.g. water) for endotoxin. A quantitative LAL test estimates the level of endotoxin in/on a product when found to be present in a gel clot limit test.
Microorganism Analysis
FDA and ISO guidance recommend characterization of the microorganisms recovered from a bioburden test or by environmental monitoring. The characterization allows for detection of change to the normal bio-load of a product, particularly if the bioburden data is used to establish a sterilization process.
SBTL offers the following to provide this information:
· Gram Stain
· Cell Morphology
· Colony Morphology
· Selective culturing for specific organisms
USP Antimicrobial Effectiveness Test
This test determines the effectiveness of an antimicrobial preservative over the life of a product. This effectiveness must be demonstrated for all injections packaged in multiple-dose containers and multiple-dose topical and oral dosage form.
USP Microbial Limits
When microbiological quality of a material is required in an individual monograph, this testing estimates quantitative levels of aerobic organisms to determine whether the material or formula meets the specifications. Five organisms are indicated for use in this testing: Staphylococcus aureus, Pseudomonas aeruginosa, Bacillus subtilis, Candida albicans, and Aspergillus niger.
Typically, these tests are performed by a membrane filtration method or the plate count method. A validation must be performed to assure the test method’s capability to detect microorganisms if present. Any change in a product’s formulation would require revalidation of the test method.
Bioburden
Definition: the population of viable microorganisms on or in a product and/or sterile barrier system.
An important part of the manufacturing process for medical devices is determining the level and nature of viable microorganisms that are generated as a result of the process. By developing and following a routine, documented sampling plan, you are assured that your process remains under control.
At SBTL, all bioburden testing is performed in a clean room, using a validated method most suited to your product following recovery methods outlined in ANSI/AAMI/ISO 11737-1. Various methods are considered; shaking, ultrasonication, stomaching, fluid pathway, membrane filtration, disintegration by blending, or swabbing are some of the methods that are reviewed to determine the optimal method for an individual product.
For medical devices, the most commonly requested information is:
· Aerobic Total Count
· Aerobic Total and Spore Count
· Mold and Fungi Total Count
· Anaerobic Total Count
· Anaerobic Total and Spore Count
Additionally, we service the personal care industry, performing bioburden estimation or screening for specific contaminants on products such as wipes, soaps, and lotions. To monitor process water and/or water systems, a Heterotrophic Plate Count and Total Coliform Count are suggested.